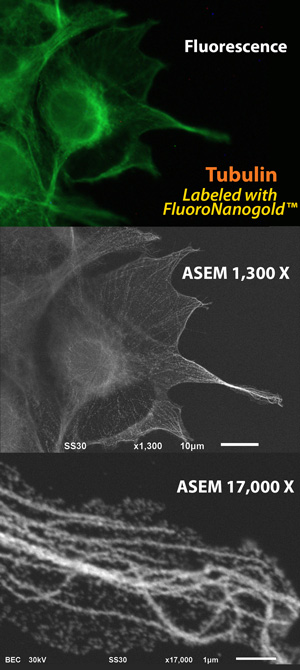
Fig. 1 Microtubules, visualized by optical microscopy (OM) and atmospheric scanning electron microscopy (ASEM: inverted SEM). COS7 cells were labeled with anti-alpha-tubulin primary antibody followed by Alexa Fluor 488 FluoroNanogold secondary antibody (Nanoprobes). Top: Fluorescence microscopy image. Middle: ASEM image of the same cells after gold enhancement by GoldEnhance EM (Nanoprobes). Botton: Higher magnification image of the right filopodia. Microtubule rails are clearly observed in the cytoplasm. (Sato et al., 2013)
Highly dynamic cellular processes are best studied in living cells, and now there's a way to watch the underlying ultrastructural changes at extremely high magnification, with a new electron microscope that works under natural conditions.
In their recent study, Dr. Chikara Sato's group and collaborators describe this new technique, which brings together optical microscopy with an "atmospheric" scanning electron microscope (ASEM). Here, cells are observed near-simultaneously both optically (using fluorescence) and at ultrastructural magnifications (using the ASEM), while resting undisturbed in standard buffer, at room temperature, and normal atmospheric pressure. A single combined probe, FluoroNanogold™, labels structures of interest for correlation between the two modalities.
Optical microscopy from above and electron microscopy from below
As Sato's group describe, cells are minimally prepared for observation, with a light microscope hovering above the specimen, while the ASEM looks up from below. The electron optics are located just under the specimen dish, which supports a silicon nitride window, coated to bind and immobilize the cells being studied. This allows electron microscopy of cells in their natural state, and avoids the changes in ultrastructure normally introduced by the dehydration and embedding of conventional electron microscopy. To keep an eye on corresponding cell dynamics at the macro level, the specimen is quasi-simultaneously observed by the optical microscope, in this case a fluorescence confocal microscope, located immediately above the specimen.
Even with such powerful techniques, identifying structures of interest requires labeling, in both fluorescence and electron microscopy. To mark their targets, the group chose FluoroNanogold™ from Nanoprobes, an immunoprobe with both a fluorescent label and a covalently bound gold nanoparticle: one probe for both microscopes. After confocal observation of the fluorescent signals from the bound probe, they then used gold enhancement with GoldEnhance™ – the catalytic deposition of metallic gold onto the gold nanoparticles – to enlarge the gold particles and render them even more easily visible by backscatter detection in the atmospheric SEM. In this way, a single label allowed exact correlation between structure and ultrastructure using the two modalities.
Correlative fluorescence and electron microscopy of moving targets: seeing the mechanism behind cellular processes and interactions
The authors evaluated their approach using over 50 different primary antibodies to study a variety of events in different primary cell cultures. Drosophila neurons were immunolabeled with anti-BP102 primary antibody followed by FluoroNanogold™ secondary; correlation of the fluorescence and electron micrographs revealed strong signals at the intra-axonal boundaries, affording a method for observing axonal segmentation. The natural reaction of mouse cerebellar and cortical neurons to magnetic beads loaded with Glutamate receptor δ2 (GluRδ2), a synapse inducer, was observed by immunostaining for presynaptic vesicular glutamine transporter1(VGluT1) and presynaptic active zone protein Bassoon. Presynaptic proteins were accumulated on the magnetic beads concentrated at the site of contact neurites, and atmospheric SEM indicated that presynaptic proteins surrounded each bead when a neurite was attached, providing a means to view some of the processes involved in synapse formation. In another experiment, phagocytosis of lactic acid bacteria by dendritic cells from mouse bone marrow was also successfully observed and analyzed.

Dr. Chikara Sato and colleagues with the Atmospheric Scanning Electron Microscope (ASEM).
By immobilizing the cells on a suitable adhesive coating, the authors achieved resolution with ASEM as high as 8 nm in specimens up to 2-3 microns thick. This bridges the gap between the cellular and the molecular scale, enabling the localization of components of dynamic processes with a precision impossible by optical microscopic methods alone. Importantly, it avoids the morphologically damaging consequences of traditional electron microscopic specimen preparation.
The use of a combined fluorescent and gold labeled immunoprobe, FluoroNanogold™, enables true correlation between the two observation modalities. The quasi-simultaneous nature of atmospheric SEM with optical microscopy, combined with the minimal preparation required, is an ideal combination to study the factors affecting dynamic phenomena in not only in biology, but also in fields such as materials and nanotechnology.
"We loved seeing these great images from Sato's group," said Dr. James F. Hainfeld, founder of Nanoprobes. "They've really opened the door for future studies into the mechanism behind cell processes that we just couldn't see until now."

Dr. Chikara Sato with his collaborators outside the Sato lab in Japan.
Learn more about the Sato Lab
Explore the new ASEM ClairScope™
developed by the Sato Lab in conjunction with JEOL
Reference:
Hirano, K.; Kinoshita, T.; Uemura, T.; Motohashi, H.; Watanabe, Y.; Ebihara, T.; Nishiyama, H.; Sato, M.; Suga, M.; Maruyama, Y.; Tsuji, N. M.; Yamamoto, M.; Nishihara, S., and Sato, C.: Electron microscopy of primary cell cultures in solution and correlative optical microscopy using ASEM. Ultramicroscopy (2013 Oct 22): in press.
Read the journal article online
* Alexa Fluor is a registered trademark of Life Technologies, Inc.