![[Nanoprobes Tech Support (14k)]](../Images/logo_tech.gif)
Updated: March 26, 2000
PALMITOYL UNDECAGOLD PRODUCT INFORMATION
Palmitoyl Undecagold
[Palmitoyl Undecagold product page]

| Product Name: |
PALMITOYL UNDECAGOLD |
| Catalog Number: |
4022 |
| Appearance: |
Orange-yellow solid |
| Revision: |
1.1 (March 2000) |
Technical Assistance Online
![[4022-PDF]](../Images/pdf.gif) Instructions (PDF) Instructions (PDF)
![[4022-PDF]](../Images/pdf.gif) Material Safety Data Sheet (PDF) Material Safety Data Sheet (PDF)
General Information
Palmitoyl-UNDECAGOLD consists of the 0.8 nm UNDECAGOLD particle1 covalently linked to a single palmitoyl molecule. Conjugation is via an amide linkage to the carboxylic group at the head of the molecule. It is intended as a lipid label for use with micelles and other dual-phase systems. Its structure is shown in figure 1:
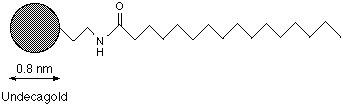
Figure 1: Structure of Palmitoyl-UNDECAGOLD (not shown to scale).
PALMITOYL-UNDECAGOLD is supplied as a solid, dried from dichloromethane solution. It should be frozen upon receipt, and stored at -20°C.
Warning: For research use only. Not recommended or intended for diagnosis of disease in humans or animals. Do not use internally or externally in humans or animals. Non radioactive and non carcinogenic.
* US Patent pending.
Instructions for Use
Palmitoyl-UNDECAGOLD is hydrophobic, and can insert into organic phases in systems such as micelles.2 It is slightly soluble in methanol and in methanol-trichloromethane and methanol-dichloromethane mixtures. Once incorporated into micelles or other structures, it may be used according to the procedures required by individual experiments in the same manner as unlabeled palmitic acid or its derivatives.
UNDECAGOLD has the following extinction coefficient at 420 nm wavelength:
| WAVELENGTH (nm) |
EXTINCTION COEFFICIENT* |
| 280 |
1.7 X 105 |
| 420 |
0.47 X 105 |
* Measured for 5 X 10-6 M solution in methanol.
General Considerations for Staining with Undecagold Reagents
Basically, normal methodologies may be used successfully with UNDECAGOLD immunoreagents. The concentration of antibody and gold is similar to other commercial preparations of colloidal gold antibodies. Therefore similar dilutions and blocking agents are appropriate.
The major difference will be in the results:
- UNDECAGOLD is an extremely uniform 0.8 nm diameter gold particle (ca.10%).
- UNDECAGOLD conjugates contain absolutely no aggregates. This is in sharp contrast to other colloidal gold conjugates that usually are prepared by centrifugation to remove the largest aggregates and frequently contain smaller aggregates.
- Close to 1 UNDECAGOLD particle to 1 lipid molecule make this product distinct from the 0.2 - 10 variable stoichiometry of colloidal gold conjugates.
- UNDECAGOLD particles do not have affinity to proteins as do other other colloidal golds. This reduces background and false labeling.
Using Stains with Undecagold
Because the 0.8 nm UNDECAGOLD particles are so small, over staining with osmium tetroxide, uranyl acetate or lead citrate will obscure direct visualization of individual UNDECAGOLD particles, and therefore these stains should not ber used. Only light staining with a low atomic number stain, such as NANOVAN, a Vanadium based negative stain, should be used.
Special Considerations for Viewing Undecagold in the Electron Microscope
UNDECAGOLD is the smallest gold probe commercially available, being just 0.8 nm in diameter. A high resolution instrument such as a Scanning Transmission Eelectron Microscope (STEM) is required for visualization; in a conventional TEM the UNDECAGOLD particles are not visible. With careful work, however, UNDECAGOLD may be seen directly in the STEM. However, achieving the high resolution necessary for this work may require new demands on your equipment and technique. Several suggestions follow:
- Before you start a project with UNDECAGOLD it is helpful to see it so you know what to look for. Dilute the UNDECAGOLD stock 1:5 in methanol and apply 4 microliters to a grid for 1 minute. Allow to dry.
- View UNDECAGOLD using a full width scan of 128 nm or less; this will give sufficient magnification for visualization.
- UNDECAGOLD is sensitive to beam damage (contrary to NANOGOLD® which is very beam-resistant); the behavior of UNDERCAGOLD in the STEM has been described in the literature3 Image at approximately 200 eÅ-2.
- In order to operate at high magnification, thin carbon film over fenestrated holey film is recommended. Many plastic supports are unstable under these conditions of high magnification/high beam current and carbon is therefore preferred. Contrast is best using thinner films.
Silver Enhancement of Undecagold for EM
UNDECAGOLD will nucleate silver deposition resulting in a dense particle 2-20 nm in size or larger depending on development time. However, silver enhancement will be slower and much less uniform than with larger gold particles such as NANOGOLD®.4 If specimens are to be embedded, silver enhancement is usually performed after embedding, although it may be done first. It must be completed before any staining reagents such as osmium tetroxide, lead citrate or uranyl acetate are applied, since these will nucleate silver deposition in the same manner as gold and produce non-specific staining.
Our LI SILVER silver enhancement system is convenient and not light sensitive, and suitable for all applications. Improved results in the EM may be obtained using HQ SILVER, which is formulated to give slower, more controllable particle growth and uniform particle size distribution.
Specimens must be thoroughly rinsed with deionized water before silver enhancement reagents are applied. This is because the buffers used for antibody incubations and washes contain chloride ions and other anions which form insoluble precipitates with silver. These are often light-sensitive and will give non-specific staining. To prepare the developer, mix equal amounts of the enhancer and initiator immediately before use. UNDECAGOLD will nucleate silver deposition resulting in a dense particle 2-20 nm in size or larger depending on development time. Use nickel grids (not copper).
The procedure for immunolabeling should be followed up to step 6 as described above. Silver enhancement is then performed as follows:
- Rinse with deionized water (2 X 5 mins).
- Float grid with specimen on freshly mixed developer for 1-4 minutes, or as directed in the instructions for the silver reagent. More or less time can be used to control particle size. A series of different development times should be tried, to find the optimum time for your experiment.
- Rinse with deionized water (3 X 1 min).
- Mount as usual.
References
- Hainfeld, J. F., in "Colloidal Gold: Principles, Methods and Applications;" M. A. Hayat, (Ed.); Academic Press, San Diego, 1989; Vol. 1, p. 413.
- Hainfeld, J. F.; Lipka, J. J., and Quaite, F. E.; J. Histochem. Cytochem., 38, 1795 (1990).
- Lipka, J. J., Hainfeld, J. F., and Wall, J. S., J. Ultrastruct. Res., 84, 120 (1983).
- Hainfeld, J. F., and Furuya, F. R. in Immunogold-Silver Staining: Principles, Methods and Applications; Hayat, M. A. (Ed.); CRC Press, Boca Raton, FL, 1995, p. 71.
|