![[Nanoprobes Tech Support (14k)]](../Images/logo_tech.gif)
Updated: January 16, 2009
GOLDENHANCE PRODUCT INFORMATION
GoldEnhanceTM EM Formulation
[GoldEnhanceTM EM Formulation product page]

| Product Name: |
GoldEnhance - EM Formulation |
| Catalog Number: |
2113 |
| Appearance: |
Colorless solution |
| Revision: |
1.5 (January 2009) |
Technical Assistance Online
![[2113-PDF]](../Images/pdf.gif) Instructions (PDF) Instructions (PDF)
![[2113-PDF]](../Images/pdf.gif) Material Safety Data Sheet (PDF) Material Safety Data Sheet (PDF)
- Introduction
- Product Information
- Gold Enhancement for EM
- Gold Enhancement for Light Microscopy
- Gold Enhancement for Immunoblots and Membrane Blots
- References
This novel, high-quality autometallographic enhancement reagent may be used in the same manner as conventional silver enhancement reagents. However, instead of depositing silver, this product selectively deposits gold onto Nanogold® particles or colloidal gold particles.1-3
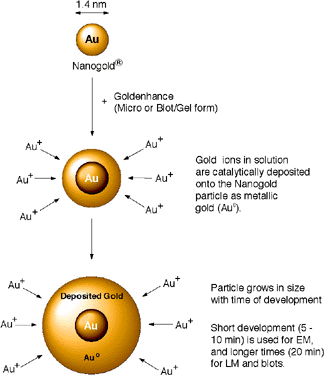
Figure 1: Enlargement of Nanogold® with GoldEnhance.
|
Why gold? Gold has several important advantages for electron microscopy, light microscopy and membrane blotting:
- Higher density; better contrast than silver.
- Improved backscatter detection of enlarged particles - more useful for SEM.
- Lower background than silver enhancement in some cases. Autonucleation is minimal even after 1-2 hours.
- Gold is not etched by osmium tetroxide: gold toning or other treatment is not necessary.
- Low viscosity for easy and accurate mixing of components.
- Milder pH conditions than silver enhancement: Goldenhance is used at near neutral pH.
- Goldenhance can be used in physiological buffers - gold is not precipitated by halides as silver is (however, rinsing with water first is still recommended).
- Excellent shelf life.
|
This reagent consists of 2 ml Solution A (enhancer), 2 ml Solution B (activator), 2 ml Solution C (initiator), and 2 mL Solution D (buffer), sufficient for up to 200 grids (using 40 microliters per grid). The reagent is formed by combining equal volumes of Solution A and Solution B, then Solution C followed by Solution D. The mixture should be prepared immediately before use. For optimum results, we recommend waiting 5-10 minutes after mixing A and B before adding C and D, although the the reagent will produce successful enhancement if C and D are added immediately or up to two hours later. Nanogold® or colloidal gold nucleates deposition of gold to give electron-dense enlarged colloidal particles in the electron microscope.
Please Note: This formulation is intended principally for electron microscopy, although it may be applicable to some light microscopy or blotting applications. The alternative LM/Blot formulation (catalog number 2112) is intended for use in light microscopy and in membrane blots.
This time period for optimum gold enhancement varies with application, but 3 to 20 minutes has been found to be optimal for enlarging the 1.4 nm Nanogold® particles to 3-20 nm or larger in size: 3-10 minutes will give particles from 3 to 20 nm in size, and longer development times will give larger particles up to 50 nm in size.
Store the component solutions in the refrigerator at 2-8°C. Avoid cross-contamination of the solutions: to prevent replacing the caps on the wrong bottles, the cap of the Solution A (enhancer) is green and that of Solution B (activator) is yellow, while that of Solution C (initiator) is purple and that of Solution D (buffer) is white. Avoid skin contact.
Warning: For research use only. Not recommended or intended for diagnosis of disease in humans or animals. Do not use internally or externally in humans or animals.
Note: All components should be equilibrated to room temperature prior to the enhancement procedure.
Contents
GoldEnhanceTM is prepared immediately before use by mixing equal amounts of Solution A (enhancer) and Solution B (activator), followed by a third equal volume of the Solution C (initiator), and a fourth equal volume of Solution D (buffer). For optimum results, we recommend waiting 5-10 minutes after mixing A and B before adding C, although the reagent will produce successful enhancement if C and D are added immediately or up to two hours later. The reagents are supplied in dropping bottles for easier dispensing of small amounts.
We recommend the use of copper or gold grids with formvar coating. We have received one report of corrosion of nickel grids by this reagent, although other users have not reported similar problems. If aldehyde-containing reagents have been used for fixation, it is recommended that these be quenched before labeling. This may be achieved by incubating the specimens for 5 minutes in 50 mM glycine solution in PBS (pH 7.4). Ammonium chloride (50 mM) or sodium borohydride (0.5 - 1 mg/ml) in PBS may be used instead of glycine.
The following procedure has been found to be effective for enhancement of tissue sections for enlargement of the 1.4 nm Nanogold®. to 3-20 nm or larger for electron microscope observation; 3-10 minutes will give particles from 3 to 20 nm in size, and longer development times will give larger particles up to 50 nm in size. However, this reagent is intended for use in a range of conditions, and different washes and development times may give better results in your system. You should follow your normal procedure up to the application of the gold conjugate; the protocol below describes the steps after this:
- Incubate with the immunogold or Nanogold® conjugate according to your usual or recommended protocol.
- Optional: postfix with 1 % glutaraldehyde in PBS.
- Wash 3 X 5 mins with PBS with 50 mM glycine (after glutaraldehyde postfix only - to remove aldehydes)
- Wash 3 X 5 mins in PBS-BSA.
- Wash 3 X 5 mins in distilled water.
- Gold enhance. Use equal amounts of the four components (Solutions A,B,C, and D); prepare about 40 µL of reagent per grid. A convenient method is to use one drop (about 10 µL) from each bottle. After mixing, a drop may be placed on a sheet of parafilm and a grid floated on it for the required time.
- First mix Solution A (enhancer: green cap) and Solution B (activator: yellow cap)
- Wait 5 min.
- Add Solution C (initiator: purple cap), then Solution D (buffer: white cap) and mix.
- Develop for the optimal particle size (usually between 3-20 min).
- Rinse with distilled water.
PBS-BSA Buffer:
- 20 mM phosphate
- 150 mM NaCl
- pH 7.40
- 0.5% BSA
- 0.1% gelatin (high purity)
Optional, may reduce background:
- 0.5 M NaCl
- 0.05% Tween 20
|
PBS Buffer:
- 20 mM phosphate
- 150 mM NaCl
- pH 7.40
|
Notes:
- Development starts with addition of Solution C (initiator), so apply to sample as soon as possible after adding C and D to minimize autonucleation background.
- Secondary GEEM solution: for slower development, substitute the following buffer for Solution D (buffer):
- 0.05 M sodium phosphate
- 0.1 M NaCl
- pH 6.1
- To obtain an especially dark signal, or for further development, develop longer or gold enhancement may be revitalized with a freshly mixed portion of GoldEnhanceTM (rinse with distilled ater between applications of GoldEnhanceTM).
- The development is not highly light sensitive, so it may be conducted under normal room lighting.
Contents
Note: we do not recommend the use of this reagent for light microscopy. Usually, stronger signals and higher signal-to-noise ratios may be obtained using the LM/Blot formulation of this reagent (catalog number 2112)4
Contents
Note: We do not recommend using this reagent for immunoblots or membrane blots, since stronger signals and better signal-to-noise ratios are usually obtained using the alternative LM/Blot formulation of GoldEnhance.5
Contents
- Hainfeld, J. F.; Powell, R. D.; Stein, J. K.; Hacker, G. W.; Hauser-Kronberger, C.; Cheung, A. L. M., and Schofer, C.: Gold-based autometallography; Microsc. Microanal., 5, (Suppl. 2: Proceedings); G. W. Bailey, W. G. Jerome, S. McKernan, J. F. Mansfield, and R. L. Price (Eds.); Springer-Verlag, New York, NY; 486-487 (1999) (Paper).
- Ackerley, C. A.; Tilups, A.; Bear, C. E., and Becker, L. E.: Correlative LM/TEM studies are essential in evaluating the effectiveness of liposome mediated delivery of the cystic fibrosis transmembrane regulator (CFTR) as a corrective therapy in a CFTR knockout mouse that develops lung disease. Microsc. Microanal., 5, (Suppl. 2: Proceedings); G. W. Bailey, W. G. Jerome, S. McKernan, J. F. Mansfield, and R. L. Price (Eds.); Springer-Verlag, New York, NY, 484-485 (1999).
- Powell, R. D.; Joshi, V. N.; Halsey, C. M. R.; Hainfeld, J. F.; Hacker, G. W.; Hauser-Kronberger, C.; Muss, W. H., and Takvorian, P. M.: Combined Cy3 / Nanogold Conjugates for Immunocytochemistry and In Situ Hybridization. Microsc. Microanal., 5, (Suppl. 2: Proceedings); G. W. Bailey, W. G. Jerome, S. McKernan, J. F. Mansfield, and R. L. Price (Eds.); Springer-Verlag, New York, NY, 478-479 (1999).
- Graf, A. H.; Cheung, A. L.; Hauser-Kornberger, C.; Dandachi, N.; Tubbs, R. R.; Dietze, O., and Hacker, G. W.: Clinical relevance of HPV 16/18 testing methods in cervical squamous cell carcinoma. Appl. Immunohistochem. Molecul. Morphol., 8, 300-309 (2000).; Tubbs, R.; Pettay, J.; Skacel, M.; Powell, R.; Stoler, M.; Roche, P., and Hainfeld, J.: Gold-Facilitated in Situ Hybridization: A Bright-Field Autometallographic Alternative to Fluorescence in Situ Hybridization for Detection of HER-2/neu Gene Amplification. Am. J. Pathol., 160, 1589-1595 (2002).
- Moeremans, M.; Daneels, G.; Van Dijck, A.; Langanger, G., and De Mey, J.: Sensitive visualization of antigen-antibody reactions in dot and blot immune overlay assays with immunogold and immunogold/silver staining. J. Immunol. Meth. 74, 353-360 (1984).
Technical Assistance Available.
For a complete list of references citing this product, please visit our References page.
Contents
|